Of the various ingredients that comprise a current-generation prosthetic limb, the point of connection to the human anatomy, the socket, is often considered the most problematic.
Attaching a Prosthetic Limb
Various issues involving the residual limb – skin infections and ulcerations, volume changes, perspiration, skin grafts, scarring, a short limb remnant and abnormal bone formation near the amputation site – present significant challenges to a positive outcome.
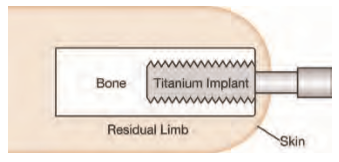
In 1990 Swedish physician Rickard Brånemark pioneered osseointegration for attaching a prosthetic limb, a technique in which a titanium bolt is surgically implanted into the medullary canal of the residual limb with one end extending percutaneously to provide an attachment point for the prosthesis. In subsequent years, teams in Europe, Asia, South America and Australia have published clinical studies on their results involving more than 200 patients, mostly transfemoral amputees but also including a significant number of upper-limb and a few transtibial patients.
The results indicate osseointegration can eliminate many problems inherent in prosthetic socket attachment for appropriate patients:
• end weight-bearing is restored;
• prosthesis control is enhanced and energy expenditure substantially reduced;
• risk of sudden prosthesis detachment from the body is minimized;
• common donning and doffing problems are resolved;
• user perception of the limb’s place in space (proprioception) is
much improved; and
• residual limb pain and skin breakdown caused by constant contact with the socket environment are virtually eliminated.


